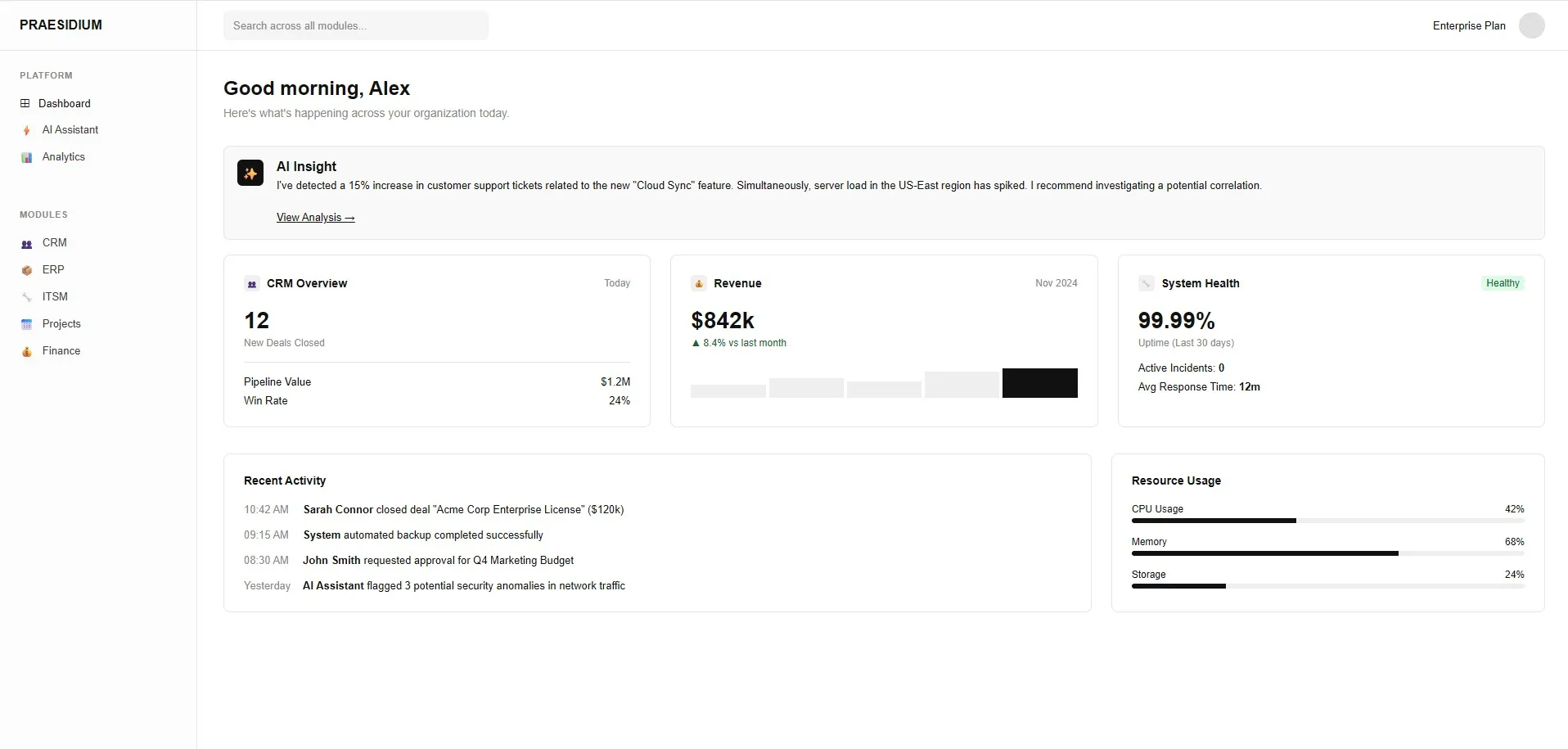
Biotech & Pharma ERP Software
Accelerate research and ensure compliance with custom Life Sciences software. Praesidium builds Biotech ERPs with 21 CFR Part 11 electronic signatures, electronic batch records (EBR), and CAPA tracking.
Consult with an Expert
Free strategy sessionBook a consultation today to discover how custom Biotech & Pharma software can reduce costs and increase efficiency.
SCHEDULE NOWIs Generic Software Like SAP Life Sciences Holding You Back?
Praesidium Builds Solutions That Fit. Tired of workarounds and disconnected tools?
Legacy Software
Custom ERP
Rigid software that limits agility.
Flexible architecture that adapts directly to your workflows.
Data scattered across multiple apps?
A unified ecosystem customized for Biotech & Pharma.
Worried about complex implementation?
Our expert team guides you from design to deployment and beyond.
We Handle the Hard Stuff.
You Focus on Growth.
Switching systems shouldn't be a nightmare. Our dedicated engineering team handles every aspect of the transition, ensuring a zero-downtime launch.
Seamless Data Migration
We securely transfer all your historical data from legacy platforms to ensure no data is left behind.
Turnkey System Setup
We configure every module, workflow, and user permission to match your exact operational needs before you log in.
White-Glove Support
We don't just send a manual. Our experts train your team to ensure everyone is confident from Day 1.
Work Smarter, Not Harder
Our platform isn't just about efficiency; it's about giving you back your time. Experience a system that anticipates your needs and handles the details.
21 CFR Part 11 Compliance
Strict audit trails, secure biometric/dual authentication, and electronic signature enforcement
Electronic Batch Records (EBR)
Digital execution of master batch records with real-time validation and deviation flagging
QMS & CAPA Management
Integrated workflow for Corrective and Preventive Actions, non-conformances, and OOS results
Cleanroom & Materials Tracking
Environmental monitoring integration and strict quarantine/release inventory protocols
Clinical Trial Accounting
Financial tracking for R&D phases, grant management, and burn-rate forecasting
Serialization & Track/Trace
DSCSA compliance with item-level serialization and supply chain traceability
Your Business,
Autopilot Enabled
Imagine a workday where reports generate themselves, inventory tracks itself, and problems are flagged before they happen. That's the power of our AI-native platform.
Common Questions
Everything you need to know about our Biotech & Pharma solutions.
What is Biotech & Pharma ERP software?
Life Sciences ERP integrates financial management, supply chain, and manufacturing with strict Quality Management Systems (QMS) required by the FDA and EMA.
Why choose Praesidium over SAP or MasterControl?
Praesidium builds custom, validated software at a fraction of the cost and implementation time of tier-1 legacy systems. We build your specific SOPs directly into the software.
Is Praesidium 21 CFR Part 11 compliant?
Yes. Our systems are built from the ground up to support FDA 21 CFR Part 11 compliance with unalterable audit trails, secure electronic signatures, and system validation documentation.
The System Modules
Everything you need to run your operations, integrated into one seamless platform.
Projected Impact
Our industry-specific solutions are designed to deliver measurable improvements across all key performance indicators.
Ready to Transform Your Operations?
Schedule a personalized demo to see how our platform can revolutionize your business
Let's Build Something Great
Tell us about your business needs and we'll create a custom solution roadmap tailored to your goals.